Multifocal rectal carcinoid tumor: a case report and literature review
Introduction
Carcinoid is a well-differentiated, indolent neuroendocrine tumor, as described by the current World Health Organization (WHO) classification system (1). Though rectal carcinoid is the second most common gastrointestinal carcinoid neoplasm (16.3%), they only comprise 1.8% of all rectal malignancies (2-5). The incidence of rectal carcinoids has increased 10-fold in the last 35 years according to the Surveillance, Epidemiology, and End Results (SEER) registry database of the National Cancer Institute, likely due to the increase in endoscopic screening (6).
Most rectal carcinoid tumors are localized and singular upon initial presentation, and treatment is based on tumor size (2,7-9). Tumors <1 cm are amenable to endoscopic resection, whereas 1–2 or >2 cm tumors may require transanal or rectal resection (10,11). Local recurrence rate after resection is low, ranging from 0–3%. An overall 5-year prognosis is high at 88.3% (2,11-13). Multifocal rectal carcinoid tumors are extremely rare, having a reported incidence of 2% to 4.5% of all rectal carcinoid tumors (7-9,14).
We report the case of a 55-year-old male who presented with rectal carcinoid tumor incidentally found on screening colonoscopy. The tumor was multifocal. The patient underwent multiple endoscopic resections. Eventually, he came to have a lower anterior resection.
Case presentation
A 55-year-old African-American male was referred to our colorectal surgery clinic by his gastroenterologist. His initial lesion was found on screening colonoscopy three years prior, when he was incidentally found to have a 3 mm sessile polyp in the rectum. Biopsy of the lesion showed well-differentiated neuroendocrine tumor, grade 1 with zero mitotic figures. Upon repeat colonoscopy 3 months later, the polyp was fully removed by SNARE polypectomy, and an additional 3mm polyp was resected and also found to be a carcinoid tumor (Figure 1). He reported no signs or symptoms of carcinoid syndrome. Normal serum calcium levels ruled out potential MEN1 syndrome.
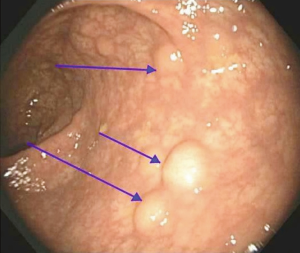
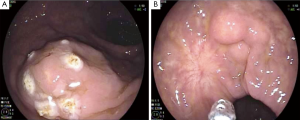
A surveillance colonoscopy 20 months later showed a 3 mm sessile polyp at the recto-sigmoid junction that was also a grade 1 carcinoid tumor. Several follow-up flexible sigmoidoscopies ensued. This allowed for SNARE polypectomy with negative margins of a 5 mm submucosal carcinoid lesion, biopsies of multiple small nodular clusters, and biopsies of four defined submucosal nodules approximately 5–10 mm in size. These submucosal nodules were located at 10–23 cm from the anal verge (Figure 2). Histology showed them all to be well-differentiated neuroendocrine tumor, grade 1, involving rectum and distal sigmoid with no mitosis or necrosis. The patient was referred for an octreotide scan and surgical consultation.
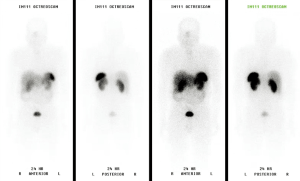
Formal colorectal surgery consultation 8 months later confirmed the prior diagnosis with endoscopic biopsies. A tattoo was placed distal to the prior biopsy scars and known nodules (Figure 3). Given the multifocal nature of his carcinoid tumor, a multidisciplinary decision was made, along with the patient, to perform a low anterior resection after full metastatic workup was completed. The octreotide scan and staging CT showed no evidence of metastatic neuroendocrine tumor (Figure 4).
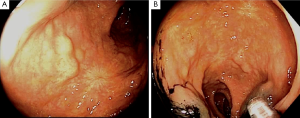
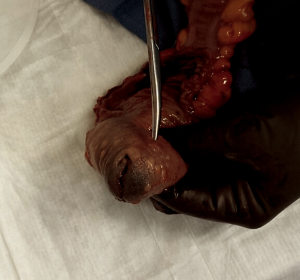
He underwent an elective robotic-assisted laparoscopic low anterior resection of his rectum for carcinoid tumor (Figure 5). Prior to the completion of the resection, flexible endoscopy was performed to ascertain the presence of any lesions distal to the tattoo. Primary anastomosis of the descending colon to the distal 1/3 of the rectum was formed, with 21 cm of colon removed without diversion. His postoperative course was relatively unremarkable, with discharge on postoperative day 4. The patient showed no signs or symptoms of carcinoid syndrome postoperatively.
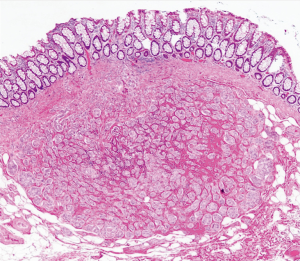
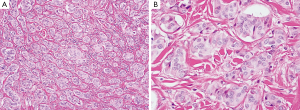
The macroscopic examination showed eight nodules ranging from 0.3 to 1.1 cm in greatest dimension. Microscopic examination revealed homogeneous nests of bland cells with abundant pink cytoplasm and bland nuclei with salt and pepper chromatin pattern and mitotic rate <2% consistent with well-differentiated neuroendocrine tumor. The tumor is extending into the submucosa (Figures 6,7). Proximal and radial margins were negative for tumor. Five out of seventeen lymph nodes were positive for carcinoid tumor with no definite lymphovascular invasion identified. One lesion was a tubular adenoma negative for high-grade dysplasia. Pathologic stage classification was pT1bpN1. PET-CT at 2 months after the procedure showed no evidence of active disease.
Discussion
This is a unique presentation of rectal carcinoid tumor due to its multifocal nature and lymph node involvement. In a review of the current literature, we found fewer than 40 cases of multifocal rectal carcinoid tumor published in the English language. Only five cases had confirmed lymph node metastasis (Table 1). Kanter et al reported a similar case of a 50-year-old African-American male with three confirmed rectal carcinoid polyps who underwent lower anterior resection and was found to have node-positive disease. At his four-year follow-up, he was not found to have recurrent or metastatic disease (21).
Table 1
| Source, year | Sex, ethnicity | Age (years) | # of lesions | Size of carcinoids (mm) | Surgical approach | Lympho node metastasis | Recurrence |
|---|---|---|---|---|---|---|---|
| Doi (15), 2016 | M, unk | 61* | 42 | <1 to 6 | Intersphincteric resection | 0/14 | 0 at 5 years |
| M, unk | 61* | 36 | <1 to 5 | Intersphincteric resection | 0/22 | 0 at 5 years | |
| Zhou (16), 2015 | M, Chinese | 47 | 3 | 5–7 | TEM | 0 | 0 with 2-year f/u |
| Sasou (17), 2012 | M, Japanese | 51 | 5 | 2.5–7 | Unk | 3/17 | 0 with 10-year f/u |
| M, Japanese | 58 | 3 | <8 | Unk | 4/6 | 0 with 1 year | |
| Ghassemi (18), 2010 | F, unk | 53 | 10–12 | 2–3 | Observation | Unk | Unk |
| Haraguchi (19), 2007 | M, unk | 69 | 30 | <10 | APR | 0 | 0 with 6 mo |
| Okamoto (20), 2004 | M, unk | 54 | 4 | <6 | ESMR | NA | Unk |
| Kanter (21), 1987 | M, African-American | 50 | 17 | <10 | LAR with Hartmanns pouch | 7/14 | 0 at 4 years |
| Maruyama (22), 1987 | M, Japanese | 52 | 5 | 4–10 | LAR | 0 | 0 |
| Scoma (23), 1978 | F, White | 58 | 3 | 2–7 | Local excision | Unk | Unk |
| Bates (14), 1962 | M, Caucasian | 51 | 3 | 2–5 | Laparotomy | Yes | Died at 13 mo |
| M, Caucasian | 56 | 2 | 10–60 | APR | No, met to liver | 0 at 15 mo | |
| M, Caucasian | 58 | 2 | 5–40 | Local excision | Yes, local excision | Died at 38 mo | |
| Saxe (24), 1964 | M, Hispanic | 52 | 2 | 10–30 | Local excision | Unk | Unk |
| Rigdon (25), 1946 | M, African-American | 60 | Unk | <10 | 0 | Deceased before resect |
*, patients were twins. unk, unknown; TEM, transanal endoscopic microsurgery; APR, abdominoperineal resection; ESMR, endoscopic mucosal resection; LAR, lower anterior resection.
Small rectal carcinoid tumors ≤10 mm treated by endoscopic resection rarely have local recurrence (0–2%), nodal involvement (2.4%) or distant metastatic disease (0.2%) (11,26,27). Our patient initially presented with a singular lesion and was later found to have additional tumors. There is no doubt that the rectal carcinoid was multifocal in nature. The question lies in whether the lesion found on his surveillance colonoscopy was a recurrence of his initial tumor or if it was a continued manifestation of his multifocal disease. There is some intrinsic risk that the initial endoscopic resection may not have completely removed the lesion (28). However, histology from the patient’s subsequent polypectomy confirmed negative margins, suggesting a low chance of residual tumor. It is possible that is was local recurrence attributable to a more aggressive tumor, especially given the positive node involvement.
Typically, carcinoid tumor size >10 mm and lymphovascular invasion are associated with the presence of nodal disease (13). Our patient’s surgical pathology showed 5 out of 17 positive nodes with no lymphovascular invasion. Although one lesion measured 11 mm, the majority of his tumors were <10 mm.
It is also interesting to note that rectal carcinoid tumors are over-represented in black and Asian populations in the USA (2). Given that our patient is African-American and the majority of the reported cases are of black or Japanese patients, there is the possibility that certain populations may be more susceptible to multifocal rectal carcinoid tumors. However, this must be observed in the context of increased incidence of rectal carcinoids in general in patients of black and Asian ethnicity. Further research into the demographics, presentation, genetics, and histology of this rare tumor are needed to clarify why this disparity exists in the USA.
Conclusions
It is important to develop a deeper understanding of multifocal rectal carcinoid tumors. Our case of a 55-year-old African-American male with multifocal recurrent rectal carcinoid tumor with nodal involvement provides is illuminating.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/dmr.2018.08.01). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Bosman FT, Carneiro F, Hruban RH, et al. WHO Classification of Tumours of the Digestive System. 4th ed. Lyon: IARC, 2010. p13-4.
- Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer 2003;97:934-59. [Crossref] [PubMed]
- Gustafsson BI, Kidd M, Modlin IM. Neuroendocrine tumors of the diffuse neuroendocrine system. Curr Opin Oncol 2008;20:1-12. [Crossref] [PubMed]
- Godwin JD 2nd. Carcinoid tumors. An analysis of 2,837 cases. Cancer 1975;36:560-9. [Crossref] [PubMed]
- Modlin IM, Sandor A. An analysis of 8305 cases of carcinoid tumors. Cancer 1997;79:813-29. [Crossref] [PubMed]
- Scherübl H. Rectal carcinoids are on the rise: early detection by screening endoscopy. Endoscopy 2009;41:162-5. [Crossref] [PubMed]
- Caldarola VT, Jackman RJ, Moertel CG, et al. Carcinoid tumors of the rectum. Am J Surg 1964;107:844-9. [Crossref] [PubMed]
- Quan SH, Bader G, Berg JW. Carcinoid tumors of the rectum. Dis Colon Rectum 1964;7:197-206. [Crossref] [PubMed]
- Orloff MJ. Carcinoid tumors of the rectum. Cancer 1971;28:175-80. [Crossref] [PubMed]
- National Comprehensive Cancer Network. Neuroendocine Tumors of the Gastrointestinal Tract, Lung and Thymus (Carcinoid Tumors) (Version 3.2017). Available online: https://www.nccn.org/professionals/physician_gls/pdf/neuroendocrine.pdf.
- Kwaan MR, Goldberg JE, Bleday R. Rectal carcinoid tumors: review of results after endoscopic and surgical therapy. Arch Surg 2008;143:471-5. [Crossref] [PubMed]
- Rodrigues Â, Castro-Poças F, Pedroto I. Neuroendocrine Rectal Tumors: Main Features and Management. GE Port J Gastroenterol 2015;22:213-20. [Crossref] [PubMed]
- Shields CJ, Tiret E, Winter DC, et al. Carcinoid tumors of the rectum: a multi-institutional international collaboration. Ann Surg 2010;252:750-5. [Crossref] [PubMed]
- Bates HR. Carcinoid tumors of the rectum. Dis Colon Rectum 1962;5:270-80. [Crossref] [PubMed]
- Doi M, Ikawa O, Taniguchi H, et al. Multiple rectal carcinoid tumors in monozygotic twins. Clin J Gastroenterol 2016;9:215-21. [Crossref] [PubMed]
- Zhou JL, Lin GL, Zhao DC, et al. Resection of multiple rectal carcinoids with transanal endoscopic microsurgery: case report. World J Gastroenterol 2015;21:2220-4. [Crossref] [PubMed]
- Sasou S, Suto T, Satoh T, et al. Multiple carcinoid tumors of the rectum: report of two cases suggesting the origin of carcinoid tumors. Pathol Int 2012;62:699-703. [Crossref] [PubMed]
- Ghassemi KA, Ou H, Roth B. Multiple rectal carcinoids in a patient with neurofibromatosis. Gastrointest Endosc 2010;71:216-8. [Crossref] [PubMed]
- Haraguchi M, Kinoshita H, Koori M, et al. Multiple rectal carcinoids with diffuse ganglioneuromatosis. World J Surg Oncol 2007;5:19. [Crossref] [PubMed]
- Okamoto Y, Fujii M, Tateiwa S, et al. Treatment of multiple rectal carcinoids by endoscopic mucosal resection using a device for esophageal variceal ligation. Endoscopy 2004;36:469-70. [Crossref] [PubMed]
- Kanter M, Lechago J. Multiple malignant rectal carcinoid tumors with immunocytochemical demonstration of multiple hormonal substances. Cancer 1987;60:1782-6. [Crossref] [PubMed]
- Maruyama M, Fukayama M, Koike M. A case of multiple carcinoid tumors of the rectum with extraglandular endocrine cell proliferation. Cancer 1988;61:131-6. [Crossref] [PubMed]
- Scoma JA. Carcinoid tumors of the rectum. Am J Surg 1978;135:708-9. [Crossref] [PubMed]
- Saxe DH. Multiple carcinoids of the rectum. Am J Surg 1953;86:553-5. [Crossref] [PubMed]
- Rigdon RH, Fletcher DE. Multiple argentaffin tumors (carcinoids) of the rectum. Am J Surg 1946;71:822. [Crossref] [PubMed]
- Gleeson FC, Levy MJ, Dozois EJ, et al. Endoscopically identified well-differentiated rectal carcinoid tumors: impact of tumor size on the natural history and outcomes. Gastrointest Endosc 2014;80:144-51. [Crossref] [PubMed]
- McConnell YJ. Surgical management of rectal carcinoids: Trends and outcomes from the Surveillance, Epidemiology, and End Results database (1988 to 2012). Am J Surg 2016;211:877-85. [Crossref] [PubMed]
- Jeon SM, Cheon JH. Rectal carcinoid tumors: pitfalls of conventional polypectomy. Clin Endosc 2012;45:2-3. [Crossref] [PubMed]
Cite this article as: Lou J, Taylor G, Arbzadeh E, Ross H. Multifocal rectal carcinoid tumor: a case report and literature review. Dig Med Res 2018;1:20.